 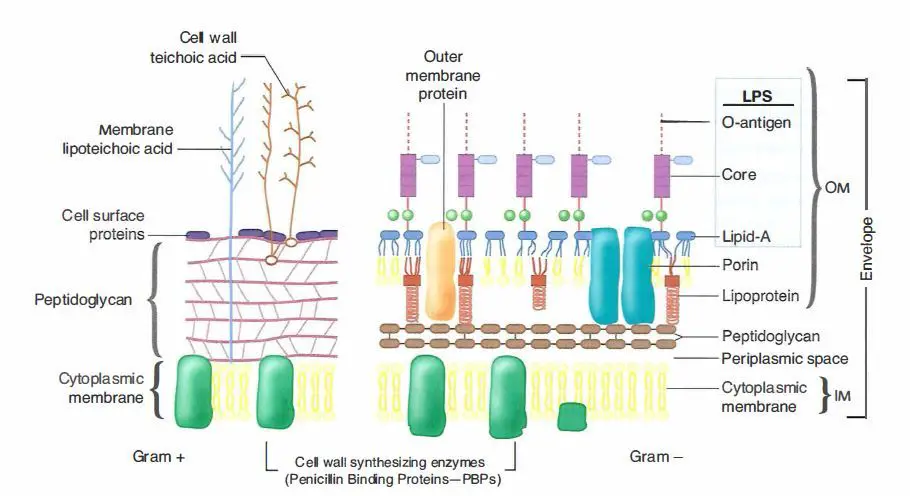
(2019) Structure and mechanism of TagA, a novel membrane-associated glycosyltransferase that produces wall teichoic acids in pathogenic bacteria. The results of this work provide insight into WTA biosynthesis and could lead to innovative approaches to treat infections caused by pathogenic bacteria.Ĭitation: Kattke MD, Gosschalk JE, Martinez OE, Kumar G, Gale RT, Cascio D, et al. As TagA enzymes are highly conserved in bacteria, they can be expected to operate through a similar mechanism. This research reveals a new method for enzyme regulation, whereby peripheral membrane association enables TagA to adopt its active form as a monomer. To understand how bacteria produce WTA, we determined the structure and deduced the enzymatic mechanism of TagA, an important enzyme that is required for WTA synthesis. aureus (MRSA) strains that lack WTA are defective in host colonization and re-sensitized to β-lactam antibiotics. The WTA biosynthetic pathway has drawn significant interest as a drug target because clinically important methicillin-resistant S. One of the most abundant polymers displayed on the surface of Gram-positive bacteria is wall teichoic acid (WTA), a negatively charged carbohydrate polymer that has critical functions in cell division, morphology, adhesion and pathogenesis. Gram-positive bacteria cause thousands of deaths in the United States each year and are a growing health concern because many bacterial strains have become resistant to commonly used antibiotics. These findings inform upon the molecular basis of WecB/TagA/CpsF activity and could guide the development of new anti-microbial drugs. 
We propose that enzyme activity is regulated by interactions with the bilayer, which trigger a structural change that facilitates proper active site formation and recognition of the enzyme’s lipid-linked substrate. italicus in its apo- and UDP-bound states reveal a novel GT fold, and coupled with biochemical and cellular data define the mechanism of catalysis. The membrane-associated TagA glycosyltransferase (GT) catalyzes the first-committed step in WTA biosynthesis and is a founding member of the WecB/TagA/CpsF GT family, more than 6,000 enzymes that synthesize a range of extracellular polysaccharides through a poorly understood mechanism. These highly abundant anionic glycopolymers have critical functions in bacterial physiology and their susceptibility to β-lactam antibiotics. Staphylococcus aureus and other bacterial pathogens affix wall teichoic acids (WTAs) to their surface. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
